Background: Unregulated handling and transfer of biological samples pose biosafety and biosecurity risks, particularly in resource-constrained research environments. This study aimed to assess the prevalence, drivers, and risk implications of unregulated biological sample handling among postgraduate students in Iraq.
Methods: A mixed-methods design was employed. Quantitative data were collected through a cross-sectional survey of 172 postgraduate students and analyzed descriptively. Qualitative insights were obtained from 25 semi-structured interviews to contextualize practices, constraints, and risk perceptions.
Results: Most participants reported difficulties obtaining samples (71.5%) and accessing adequately equipped university laboratories (78.5%), which contributed to reliance on commercial laboratories (65.1%) and informal peer-to-peer sample exchange (51.7%). Despite high awareness of biosafety and biosecurity risks (91.3% of survey respondents; 16/25 interviewees), these practices persisted due to structural constraints, including material shortages and inadequate laboratory infrastructure (reported in 21 interviews). Interviews further highlighted limited access to adequate, practical biosafety training (reported in 15 interviews). The absence of specialized governmental biosecurity oversight mechanisms was perceived to exacerbate risks related to loss of traceability, contamination, and potential misuse of biological materials.
Conclusions: Unregulated sample handling among postgraduate students in Iraq is driven primarily by systemic constraints rather than risk ignorance. Strengthening laboratory capacity, formalizing regulated pathways for sample transfer, and implementing mandatory, practical biosafety and biosecurity training alongside clearer national oversight are essential to mitigate biosafety and biosecurity risks and protect research integrity and public health.
Keywords: Biosafety, Biological Samples, Unregulated Risks, Students
Biological samples, such as tissues, blood, and bodily fluids, serve as the cornerstone of scientific advancement and innovation across medicine, pharmacology, and life sciences. The safe and regulated handling of these samples is paramount not only for ensuring the integrity of scientific research and the accuracy of its findings, but also for safeguarding the health and safety of individuals who handle them, and the wider community [1,2]. Any lapse in biological sample management practices poses a potential risk, ranging from contamination and data loss to direct health hazards associated with exposure to pathogenic agents [3,4]. The concepts of Biosafety and Biosecurity are gaining increasing prominence within academic and research environments, particularly in universities conducting extensive research involving sensitive biological materials [5]. Biosafety encompasses the principles, technologies, and practices implemented to prevent unintentional exposure to pathogens or toxins, or their release into the environment [3]. Conversely, Biosecurity focuses on protecting valuable or dangerous biological materials from theft, diversion, misuse, or intentional release [6]. Unregulated practices, or those lacking adequate oversight, directly threaten both concepts, creating an environment where samples, personnel, and the community are put at risk. These risks may become particularly pronounced in academic research environments where access to adequate laboratory infrastructure and training resources is limited.
In many research settings, especially in developing countries or those facing infrastructural challenges, postgraduate students, who constitute a vital part of the research workforce, may encounter significant limitations in accessing adequate laboratory resources and appropriate training [7]. These challenges can compel them to adopt informal or unregulated practices in handling biological samples, such as relying on commercial laboratories, exchanging samples with colleagues, or even working in inadequately equipped environments [8]. While such practices may be driven by the necessity to complete research, they open the door to serious risks concerning sample integrity loss, contamination, mismanagement, and potential exposure to infectious agents [9].
Iraq, as an academic and research environment with unique challenges, serves as a prominent example of a context where such practices can emerge. Following decades of conflict and limitations on investment in research infrastructure [10], Iraqi academic institutions may be in urgent need of a comprehensive assessment of biological sample handling practices, particularly among postgraduate students who often operate under significant time and financial pressure [11]. The current scientific literature notably lacks comprehensive studies exploring this phenomenon specifically within the Iraqi context. Therefore, there is a need for systematic investigation of how postgraduate students manage biological samples in practice and what factors drive unregulated handling behaviors.
Building on the foregoing, this study aims to bridge this knowledge gap by comprehensively assessing the risks associated with the unregulated handling of biological samples among postgraduate students in Iraq. By employing a mixed-methods design that integrates both quantitative and qualitative data, this research seeks to provide an in-depth understanding of the prevalence of these practices, the underlying reasons, students' risk perceptions, their level of biosafety awareness, and the overall impact on biosecurity. The anticipated findings will underscore the urgent need for developing effective policies and training programs to enhance biosafety and biosecurity practices within Iraqi academic institutions. This study provides empirical evidence on biological sample handling practices among postgraduate students in Iraq and highlights the structural factors contributing to unregulated practices.
This research employed a mixed-methods design to provide a comprehensive understanding of the unregulated handling of biological samples among postgraduate students in Iraq. The study combined descriptive quantitative analysis, conducted through an online questionnaire, with qualitative analysis, derived from semi-structured interviews. This mixed-methods approach allowed the study to combine quantitative evidence on the prevalence of practices with qualitative insights explaining the underlying structural and contextual factors influencing these behaviors.
Data were collected using both quantitative and qualitative instruments, as follows:
An online questionnaire was developed to gather quantitative data. It comprised four main sections: (1) Demographics, (2) Sample handling practices, (3) Risk assessment, and (4) Training and awareness. To ensure the reliability of responses, a trap question was included to detect inattentive or random answers. The instrument's content relevance was further ensured through validation by subject matter experts.
Semi-structured interviews were conducted to gather in-depth qualitative insights. This approach balances a pre-determined set of topics or open-ended questions with the flexibility to explore emerging themes and follow up on participants' responses in depth. Participants were selected using a quasi-purposive sampling method, focusing on postgraduate students with relevant experience or knowledge in biological sample handling practices. To maintain participant comfort and privacy, interviews were not audio-recorded. Instead, the researcher manually documented responses during the interviews and immediately reviewed them afterward to ensure accuracy and completeness.
A non-probability sampling approach was utilized for both components of the study:
Quantitative Component: A convenience sample of postgraduate students was recruited from various medical and life sciences fields across several Iraqi universities. Proportional representation was not a requirement for this component.
Qualitative Component: Participants for the interviews were selected using a quasi-purposive sampling method, based on their direct relevance to the research topic and their willingness to share their experiences.
Different analytical approaches were applied to the quantitative and qualitative datasets:
Quantitative Data Analysis: Quantitative data were analyzed solely using descriptive statistics, specifically frequencies and percentages. Inferential statistical tests were not applied due to the imbalanced representation of universities within the sample, which could introduce bias into the conclusions.
Qualitative Data Analysis: Qualitative data, derived from the interview responses, were analyzed using thematic analysis. This approach facilitated the identification of recurring themes and key insights pertinent to the research objectives.
Participants were informed of the study’s purpose and assured full confidentiality. Participation was voluntary, with the right to withdraw at any point, and no personally identifiable information was collected or recorded. The study was reviewed and approved by the Research Ethics Committee at the Genetic Engineering and Biotechnology Institute for Postgraduate Studies / University of Baghdad.
This section presents the findings derived from the study, which employed a mixed-methods approach. Quantitative data were collected via a survey administered to 172 postgraduate students and analyzed using descriptive statistics (frequencies and percentages). Concurrently, qualitative data were gathered through 25 semi-structured interviews and analyzed using thematic analysis to identify key themes. This chapter will first cover the quantitative demographic characteristics, followed by the survey results, and finally, the qualitative themes extracted from the interviews, to provide a comprehensive understanding of the topic.
The quantitative data collected from 172 postgraduate students across various Iraqi universities provides valuable insights into the challenges, practices, and perceptions surrounding the handling and exchange of biological samples. The findings are presented below according to key thematic areas of the study:
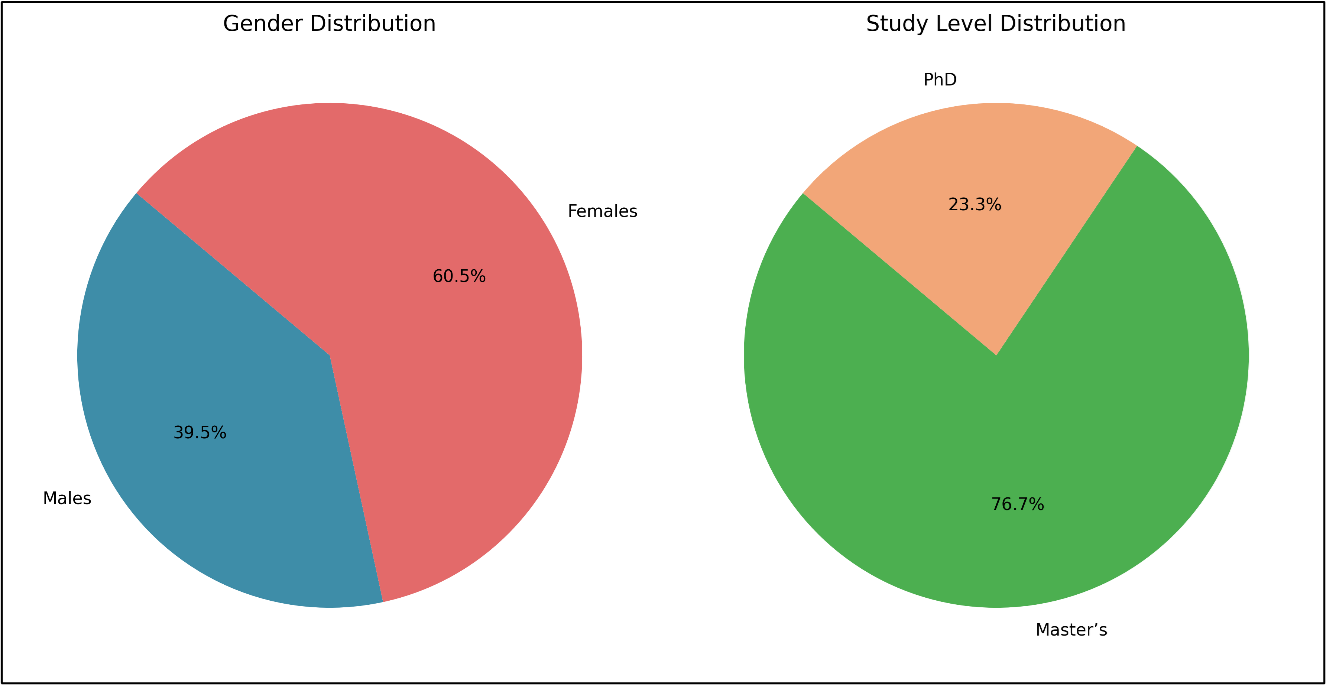
To provide a comprehensive understanding of the sample, the demographic characteristics of the participants were analyzed. Figure 1 clearly illustrates the sample's distribution in terms of gender and study level.
Regarding gender, females constituted the majority of participants, representing 60.5%, while males accounted for 39.5%. As for study level, Master's students formed the largest proportion of the sample at 76.7%, with PhD students making up 23.3%.
Figure 1: illustrates the sample's distribution by gender and study level.
The results indicate that researchers encounter significant challenges in their research endeavors. As presented in Table 1, the majority of participants (71.5%) reported facing difficulty in obtaining biological samples to complete their research. Furthermore, 78.5% of respondents indicated challenges in completing their research due to time constraints or limited access to well-equipped university labs, with an additional 20.3% encountering these issues sometimes.
Table 1: Challenges Faced by Researchers in Sample Acquisition and Lab Access
| Question | Answer Options | Count | Percentage (%) |
| Have you ever faced difficulty obtaining biological samples to complete your research? | Yes | 123 | 71.5 |
| No | 5 | 2.9 | |
| Sometimes | 44 | 25.6 | |
| Do you face difficulty completing your research due to time constraints or limited access to well-equipped university labs? | Yes | 135 | 78.5 |
| No | 2 | 1.2 | |
| Sometimes | 35 | 20.3 |
Findings revealed a significant reliance on external laboratories and sample exchange among colleagues (see Table 2, Figure 2). 65.1% of researchers reported using commercial labs or external offices for their experiments, while 80.2% had transferred samples from university labs to these external facilities to complete their research. The results also showed that 51.7% of participants had exchanged samples with colleagues to aid their research.
Table 2: Researchers' Practices in Sample Usage, Transfer, and Exchange
| Question | Answer Options | Count | Percentage (%) |
| Do you use commercial labs or external offices to conduct your experiments or tests? | Yes | 112 | 65.1 |
| No | 12 | 7 | |
| Sometimes | 48 | 27.9 | |
| Have you ever transferred samples from university labs to commercial labs or external offices to complete your research? | Yes | 138 | 80.2 |
| No | 1 | 0.6 | |
| Sometimes | 33 | 19.2 | |
| Have you ever exchanged samples with colleagues to help them complete their research? | Yes | 89 | 51.7 |
| No | 50 | 29.1 | |
| Sometimes | 33 | 19.2 |
Figure 2: Prevalence of external laboratory use, sample transfer, and peer sample exchange among postgraduate students.
The survey demonstrates a high level of awareness regarding the risks associated with sample handling (see Table 3, Figure 3). An overwhelming majority of participants (91.3%) were aware that transferring samples between commercial labs or students might pose risks to sample integrity or result accuracy. Furthermore, 60.5% of researchers expressed concern about potential leakage or loss of samples during transport or exchange. Moreover, 82.0% of respondents categorized the exchange of samples between students and commercial labs as 'high risk,' while 60.5% believed that commercial labs might share their research samples with other students.
Table 3: Researchers' Awareness and Concerns Regarding Sample Handling Risks
| Question | Answer Options | Count | Percentage (%) |
| Are you aware that transferring samples between commercial labs or students might pose risks to sample integrity or result accuracy? | Yes | 157 | 91.3 |
| No | 3 | 1.7 | |
| Maybe | 12 | 7 | |
| Have you ever felt concerned about potential leakage or loss of samples during transport between labs or exchange among students? | Yes | 104 | 60.5 |
| No | 30 | 17.4 | |
| Maybe | 38 | 22.1 | |
| What level of risk do you associate with exchanging samples between students and commercial labs? | High risk | 141 | 82 |
| Medium risk | 15 | 8.7 | |
| Low risk | 15 | 8.7 | |
| No risk | 1 | 0.6 | |
| Do you believe commercial labs may share your research samples with other students? | Yes | 104 | 60.5 |
| No | 12 | 7 | |
| Maybe | 56 | 32.6 |
Figure 3: Risk awareness versus continued risky practices among postgraduate students.
Regarding training in sample handling, findings revealed that 70.9% of participants had received practical training in biosafety within their universities (see Table 4, Figure 4). Of these, 55.2% received training during their undergraduate studies, 10.5% during postgraduate studies, and 29.1% through special courses. However, 41.3% of respondents reported a lack of written instructions or clear protocols for handling biological samples. A substantial 64.0% believed that some students begin working with samples without prior training. Concerning future support, the results indicated strong endorsement for mandatory training programs, with 69.8% of participants agreeing on the necessity of implementing such programs before allowing any student to handle biological samples.
Table 4: Researchers' Perceptions and Practices Concerning Training and Protocols
| Question | Answer Options | Count | Percentage (%) |
| Have you ever received practical training within your university on how to handle biological samples as part of biosafety and biosecurity courses? | Yes | 122 | 70.9 |
| No | 46 | 26.7 | |
| No, because my field is different | 4 | 2.3 | |
| If you received training in biosafety, where did it take place? | During undergraduate (BSc) | 95 | 55.2 |
| During postgraduate studies | 18 | 10.5 | |
| Special courses | 50 | 29.1 | |
| I didn't receive training because my field is different | 9 | 5.2 | |
| Were written instructions or clear protocols provided for handling biological samples? | Yes | 50 | 29.1 |
| No | 71 | 41.3 | |
| Maybe | 51 | 29.7 | |
| Do you think some students start working with samples without prior training? | Yes | 110 | 64 |
| No | 13 | 7.6 | |
| Maybe | 49 | 28.5 | |
| Would you support a mandatory training program before allowing any student to handle biological samples? | Yes | 120 | 69.8 |
| No | 15 | 8.7 | |
| Maybe | 37 | 21.5 |
Figure 4: Training exposure, availability of written protocols, and support for mandatory biosafety training.
This section presents the findings from the qualitative phase of the study, which involved 25 semi-structured interviews with postgraduate students. These students were drawn from diverse biological and medical science disciplines across various Iraqi universities. The interviews aimed to comprehensively explore the realities of biological sample handling, including motivations and challenges, awareness of associated risks, and the current state of biosafety training. The data were analyzed using Thematic Analysis to identify recurring patterns and insights from participants' responses.
Analysis of participants' responses revealed several consistent reasons driving the exchange of samples between students and commercial laboratories. These reasons primarily stemmed from systemic and resource-related limitations within the university environment: Time constraints and difficulties in completing research within the stipulated period were significant factors, articulated in 17 out of 25 interviews. Prohibitions on sample removal from hospitals, enforced by Ministry of Health regulations, were cited in 14 interviews, compelling students to seek external solutions.
Poor infrastructure in university laboratories and a critical shortage of essential materials emerged as a dominant concern, mentioned in 21 interviews. Reliance on commercial laboratories for quick results or the purchase of ready-made samples was also a common practice, noted in 19 interviews.
A typical sentiment encapsulating these challenges was expressed by a postgraduate student from the University of Baghdad:
"We don’t have the necessary materials, and the university labs are barely functioning for a few hours during official hours-if the equipment even works at all-so we have no choice but to go to commercial labs."
This quote underscores the direct link between inadequate university resources and the necessity of external lab reliance.
The interviews also unveiled a prevalent, albeit informal, practice of sample exchange among students. Some participants openly acknowledged sharing samples to help colleagues or to take advantage of previously examined specimens, especially in molecular research where time or resources may be scarce. As an example, one participant said:
"We don't have time to start from scratch, so we take samples from colleagues who have already finished the practical part."
Others described this as a form of "informal exchange" primarily occurring within student groups, often driven by intense time pressure and the scarcity of available samples. Overall, 17 out of 25 participants admitted to personally engaging in sample exchange, while the remaining participants were aware of others doing so.
Participants' responses regarding awareness of biological risks varied, ranging from partial acknowledgment to a notable lack of concern. While 16 participants recognized the inherent biological risks, they often viewed sample exchange as an unavoidable reality within their research context. Some even expressed a belief that the risk was "exaggerated" if samples were handled with "care." This perspective was highlighted by a student from the University of Karbala:
"I know it’s wrong, but I have no choice, and time is running out."
This statement reflects the tension between perceived risk and practical exigencies.
A critical finding was the widespread admission by most students that they had not received proper biosafety training. Specifically, 15 interviews indicated a distinct lack of clear or structured training programs. When training did exist, it was often described as: Purely theoretical, lacking practical components. Outdated, with participants having received it during their undergraduate years. Non-specialized, failing to address specific risks pertinent to their current research. One participant's statement clearly articulated this deficiency:
"The training was just a lecture in undergrad, with no practical component. We never learned how to handle dangerous samples properly."
Participants offered several key recommendations aimed at improving sample management and biosafety practices within the academic research environment: Establishing official exemptions for postgraduate students to transfer samples under clearly defined protocols.
Improving university laboratories through better infrastructure, adequate supply of essential materials, and the implementation of an on-call lab system to extend operational hours.
Developing a national training program on biosafety and biosecurity to ensure standardized and comprehensive education. Providing students with legal and practical flexibility during the execution of their research, acknowledging the complexities of fieldwork and sample acquisition.
This mixed-methods study aimed to assess the risks associated with the unregulated handling of biological samples among postgraduate students in Iraq. The integrated quantitative and qualitative findings offer a comprehensive understanding of the field's realities, illuminating existing challenges, prevalent practices, risk perceptions, and their subsequent implications for biosafety and biosecurity.
The quantitative results indicated that a substantial majority of participants (71.5% out of 172 students) faced difficulties in obtaining necessary biological samples, while 78.5% reported that time constraints and limited access to adequately equipped university laboratories hindered their research progress. These figures are deeply contextualized and elaborated by the qualitative analysis. Interviews revealed that "poor infrastructure in university labs and shortage of essential materials" was the most frequently cited reason (mentioned in 21 out of 25 interviews), directly compelling students to seek alternative solutions. A typical quote from a postgraduate student at the University of Baghdad encapsulates this reality: "We don't have the necessary materials, and the university labs are barely functioning for a few hours during official hours-if the equipment even works at all-so we have no choice but to go to commercial labs."
This resource deficit directly translates into reliance on "commercial laboratories and external offices to conduct experiments" (quantitatively acknowledged by 65.1% of students) and the transfer of samples to these external facilities (80.2% quantitatively). The absence of adequate infrastructure is not merely an academic impediment; it represents a critical biosafety vulnerability [5,12]. Once biological samples leave a controlled laboratory environment, they become susceptible to multiple risks, including contamination, loss, or unauthorized use [13]. The lack of established protocols for transferring these samples between institutions, as evidenced by these practices, compromises the chain of custody, potentially leading to inaccurate research outcomes and increasing the likelihood of exposure to hazardous biological agents outside formal oversight [14], This unregulated movement of samples also raises biosecurity concerns, as it bypasses institutional controls designed to prevent the misappropriation or misuse of sensitive biological materials [15,16].
According to quantitative data, 51.7% of students acknowledged sharing biological samples with their peers, and another 19.2% did so on occasion. This suggests that informal collaboration is a coping mechanism. This behavior is supported by qualitative data, as 17 out of 25 participants personally admitted to exchanging samples, while the remaining participants said they knew others who did so. Statements like "We take samples from colleagues who already completed the practical part, because we don't have time to start from scratch" demonstrate how students are compelled to find these easy fixes due to time constraints and a lack of available samples.
While seemingly innocuous from the students' perspective, this practice poses significant biosafety and biosecurity risks. Informal exchange means a complete absence of traceability records, making it difficult to pinpoint the sample's origin in cases of contamination or misuse [17,18]. It also elevates the risk of pathogen transmission among individuals if stringent safety procedures (e.g., proper personal protective equipment use, safe waste disposal) are not followed [19,20]. From a biosecurity standpoint, this signifies a loss of control over potential biological agents, which could lead to undeclared laboratory incidents or even the unintentional release of pathogens into the community, thereby threatening public health [19,21].
The discrepancy between high-risk awareness and ongoing participation in risky behaviors is a startling paradox in this study. 91.3% of students acknowledged, quantitatively, that sample transfer would jeopardize sample integrity and result accuracy, while 82% said sample exchange involved "high risk." However, 60.5% of respondents still engaged in these procedures despite expressing concern about possible sample loss or leakage.
Qualitative findings elucidate this cognitive and behavioral dissonance. While 16 participants acknowledged the biological risks, they perceived them as an "unavoidable reality." A student from the University of Karbala vividly expressed this dilemma: "I know it’s wrong, but I have no choice, and time is running out." This highlights that awareness alone is insufficient to alter behaviors if viable alternatives are not available. In terms of biosafety, this means students' knowledge of risks does not translate into safe practices due to academic and institutional pressures, creating a dangerous gap between awareness and the actual implementation of biosafety measures [22]. This significantly increases the likelihood of serious biological incidents, given inadequate training and protocols [21,23].
Although 70.9% of students reported receiving some form of practical instruction, qualitative results revealed the insufficient nature of this training. Fifteen interviews indicated a lack of clear training programs, with existing training described as "purely theoretical, outdated (from undergraduate years), or non-specialized." One participant elaborated: "The training was just a lecture in undergrad, with no practical component. We never learned how to handle dangerous samples properly."
This issue is compounded by the scarcity of clear written protocols for biological sample handling, confirmed by only 29.1% of students. These gaps in training and protocols pose a substantial biosafety and biosecurity risk [11,24]. The absence of specialized practical training means students may lack fundamental skills for safe sample handling, increasing the risk of exposure to pathogens, cross-contamination, and infection spread [4,25]. Furthermore, the lack of standardized protocols fosters disorganization, makes sample tracking difficult, and creates an environment conducive to biological incidents and the potential misuse of biological materials [26,27]. The strong support (69.8%) for implementing a mandatory training program underscores students' recognition of this critical gap and the urgent need for institutional intervention.
This mixed-methods study demonstrates that unregulated biological sample handling among postgraduate students in Iraq is widespread and driven primarily by systemic constraints, including limited laboratory infrastructure, material shortages, and time pressures. Despite high awareness of biosafety and biosecurity risks, students continue to rely on commercial laboratories and informal peer-to-peer sample exchange, creating vulnerabilities related to contamination, loss of traceability, and potential misuse of biological materials. The findings highlight a critical gap between risk awareness and safe practice, exacerbated by insufficient practical training and the absence of standardized protocols. Addressing these challenges requires coordinated institutional and national interventions, including strengthening laboratory capacity, formalizing regulated pathways for sample transfer under clear protocols, and implementing mandatory, practical biosafety and biosecurity training for postgraduate students. Such measures are essential to mitigate biosafety and biosecurity risks and to safeguard research integrity and public health.
1. Budimir D, Polasek O, Marusic A, Kolcic I, Zemunik T, Boraska V, et al. Ethical aspects of human biobanks: a systematic review. Croat Med J. 2011;52(3):262–279. doi:10.3325/cmj.2011.52.262
2.World Health Organization. Laboratory biosafety manual. 3rd ed. Geneva: World Health Organization; 2004.
3. Ragavan GK, Selvaraj P, Murugesan D, Krishnaswamy Gopalan T, Chinnaiyan S, Gopal DR, Ramasamy Parthiban AB, Kumaragurubaran K, Palavesam A. Management practices and technologies for efficient biological sample collection from domestic animals with special reference to Indian field conditions. Anim Dis. 2023;3(1):34. doi:10.1186/s44149-023-00096-6
4. Sveinbjornsson BR, Gizurarson S. Biohazards. In: Handbook for laboratory safety. Elsevier; 2022. p. 85–99. doi:10.1016/C2021-0-01385-7
5. Morse SA. Grand challenge in biosafety and biosecurity. Front Bioeng Biotechnol. 2025;12:1538723. doi:10.3389/fbioe.2024.1538723
6. Artika IM, Ma’roef CN. Current laboratory biosecurity for handling pathogenic viruses. Am J Biochem Biotechnol. 2018;14(2):108–116. doi:10.3844/ajbbsp.2018.108.116
7. Badr MZ. Challenges facing scientific research in developing countries: environment and resources. Egypt J Basic Clin Pharmacol. 2018;8. doi:10.11131/2018/101388
8. Verstuyft C, Antoun Z, Deplanque D, Barau C, Bordon-Pallier F, Belon F, Bertoye PH, Bilbault P, Chevalier MP, David M, Demerville L, Di Donato JH, Echard E, Villeroche VD, Lang M, Lanta M, Persoz C, Piga N, Pol S, et al. Collection of human biological samples for research purpose: key challenges and patients’ perspectives. Therapies. 2018;73(1):73–81. doi.org/10.1016/j.therap.2018.01.001
9. Chauhan A, Jindal T. Good microbiological laboratory practices. In: Chauhan A, Jindal T, editors. Microbiological methods for environment, food and pharmaceutical analysis. Cham: Springer; 2020. p. 15–22. doi:10.1007/978-3-030-52024-3_2
10. Dakhil ZA, Skuk M, Al-Jorani M. Challenges faced by Iraqi academics in career advancement and promotion: a survey-based study. Learn Teach. 2024;17(1):107–121. doi:10.3167/latiss.2024.170106
11. Alrasheed RS, Al-Mahrous MM, Alshahrani RM, Alsaadi NR. Safety and decontamination procedures for infectious sample handling. J Healthc Sci. 2024;4(12):1010–1015. dx.doi.org/10.52533/JOHS.2024.41249
12. Moritz RL, Gillum DR. Adaptation of research infrastructure to meet the priorities of global public health. Front Bioeng Biotechnol. 2021;8:613253. doi.org/10.3389/fbioe.2020.613253
13. Beeckman DSA, Rudelsheim P. Biosafety and biosecurity in containment: a regulatory overview. Front Bioeng Biotechnol. 2020;8:650. doi:10.3389/fbioe.2020.00650
14. Kane BT, Meaney JF, Luz SF. Sharing patient radiological imaging studies and pathological samples: standards, protocols and practices. In: Proc Int Workshop Managing Interoperability and Complexity in Health Systems; 2011. p. 71–74. doi.org/10.1145/2064747.2064763
15. Abad X. Transfer of biological samples from a biosafety level 3 facility. Biosafety. 2012;1(6). doi:10.4172/2167-0331.1000e125
16. Rainer D, Cook S. Overcoming regulatory gaps in biological materials oversight by enhancing IBC protocol review. In: Ensuring national biosecurity. Elsevier; 2016. p. 73–91. doi.org/10.1016/B978-0-12-801885-9.00005-6
17. Smith JA, Sandbrink JB. Biosecurity in an age of open science. PLoS Biol. 2022;20(4): e3001600. doi.org/10.1371/journal.pbio.3001600
18. Ganesh GV, Harithpriya K, Umapathy D, Hoque ME, Ramkumar KM. Laboratory safety. In: Ramkumar KM, Senthilkumar R, Hoque ME, editors. Advanced mammalian cell culture techniques. Boca Raton: CRC Press; 2023. p. 11–14. doi.org/10.1201/9781003397755
19. Golovko AM, Napnenko OO. Biological safety and biosecurity—the basis for countering new biological threats and challenges. Vet Med Inter-Dept Sci Collect. 2024;110:5–8. doi:10.36016/VM-2024-110-1
20. Jalali F, Pahlavan B, Mosayyebi M, Salehi Salut E. Identifying and evaluating occupational hazards of infectious waste landfill workers by the JHA method. J Adv Environ Health Res. 2024;12(2):116–121. doi: 10.34172/jaehr.1334
21. Blacksell SD, Summermatter K, Masuku ZM, Kojima K, Ross E, Harper DR, Hamilton K. Investment in biosafety and biosecurity: the need for a risk-based approach and systematic reporting of laboratory accidents to mitigate laboratory-acquired infections and pathogen escapes. Lancet Microbe. 2023;4(11): e854–e855. doi:10.1016/S2666-5247(23)00288-4
22. Thi Ngoc Ha Bui, Nguyen TD, Joshi TTX, Nguyen TTH. Biosafety knowledge and perception among medical laboratory students: a cross-sectional study at a medical university in Vietnam. Int J Occup Saf Health. 2024;14(1):7–16. doi.org/10.3126/ijosh.v14i1.56395
23. Padde JR, Akiteng W, Edema W, Mahjub Atiku S, Tibyangye J, Tekakwo J, Andruga C, Hope D, Musinguzi B, Gesa JB, Amadile L, Agondua R. Assessment of biosafety and biorisk management practices among medical laboratory students in two institutions in Uganda. Biosaf Health. 2022;4(6):399–405. doi:10.1016/j.bsheal.2022.08.005
24. Laboratory Specialist, Ministry of Health, Saudi Arabia; Alansari FT. Assessing laboratory safety protocols: enhancing practices for a safer environment. J Med Sci Clin Res. 2023;11(12):27–31. doi:10.18535/jmscr/v11i12.06
25. Azaev M, Kosogova T, Agafonov A, Netesov S. Fundamentals of biological safety. Moscow: INFRA-M Academic Publishing; 2023. dx.doi.org/10.12737/2001724.
26. Samuel LP. Harmonization of microbiology processes and standards: work in progress. Clin Chem Lab Med. 2018;56(10):1624–1628. doi.org/10.1515/cclm-2017-1074
27. Verma A, Yadav SK. Laboratory hazards. In: Yadav SK, Gupta R, Singh S, editors. Clinical laboratory management. Cham: Springer Nature; 2023. p. 199–210. doi:10.1007/978-3-031-46420-1_35